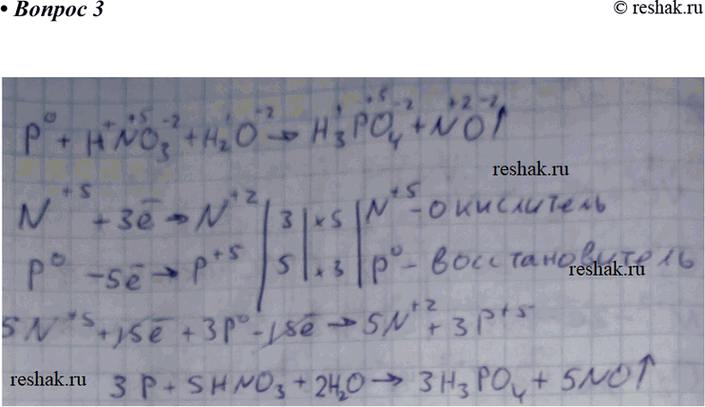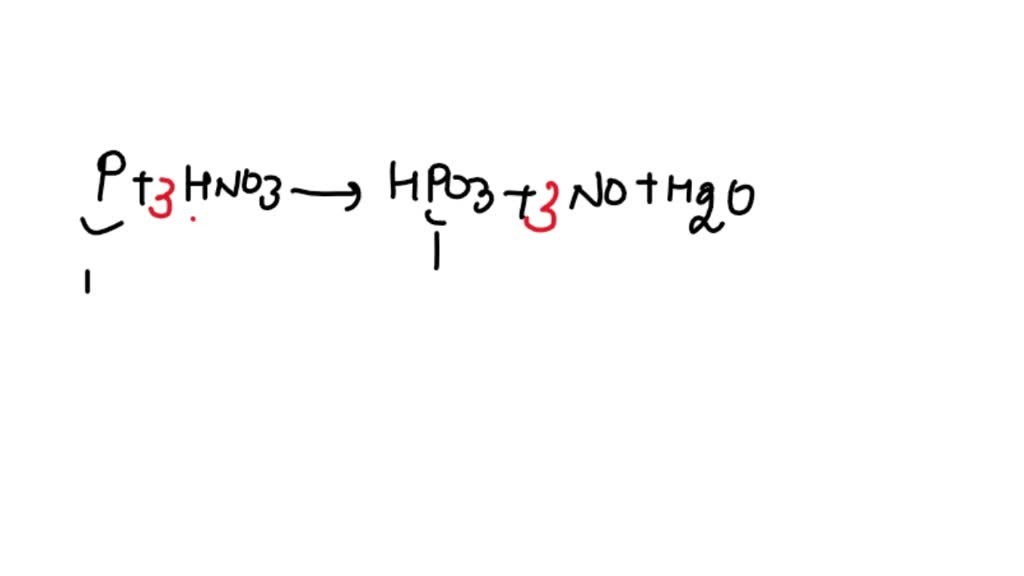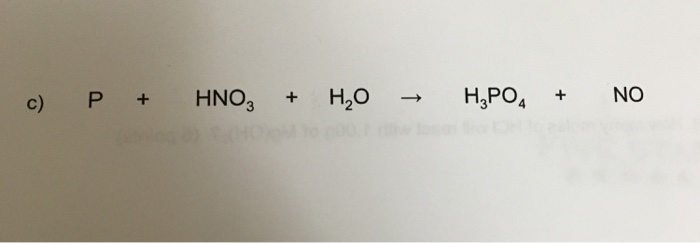When aniline reacts with nitric acid in the presence of acetic anhydride, it gives p-nitroaniline only. Why? - Quora

Concentrated nitric acid oxidised phosphorus to phosphoric acid according to the following equation: P+5HNO3(conc.)---> H3PO4 +H2O +5NO2 If 9.3 g of phosphorus was used in the reaction, calculate: I) Number of moles

Balance the chemical equation by ion electron and half reaction method. P4+ HNO3=H3PO4+NO2+H2O - YouTube

HNO3/H3PO4–NANO2 mediated oxidation of cellulose — preparation and characterization of bioabsorbable oxidized celluloses in high yields and with different levels of oxidation - ScienceDirect
Concentrated nitric acid oxidises phosphorus to phosphoric acid according to the following equation: P +5HNO3 → H3PO4 +5NO2+ H2O If 6.2 g of phosphorus was used in the reaction, calculate: a The

Используя метод электронного баланса, расставьте коэффициенты в уравнении реакции, схема которой P - Школьные Знания.com