
SOLVED: A suggested mechanism for the decomposition of ozone is: O3 0z + 0 (fast) 02 + 0 O3 (fast) 0+O3 202 (slow) What is the rate law for this reaction from

Formation of O3 in the troposphere. NO2 is cleaved by sunlight to NO• +... | Download Scientific Diagram

Amazon.com: Enerzen O-UVC3 - HEPA + UV Light + 40,000 mg/h Industrial Ozone Generator for Eliminating Odors - O3 Machine Air Ionizer Deodorizer for Home, Room, Smoke, Car, Pet : Home & Kitchen

SOLVED: A possible mechanism for the decomposition of Ozone is: O3 = 02 + 0 with forward and reverse rate constants k1 and k1 0 + O3 + 2 02 with rate
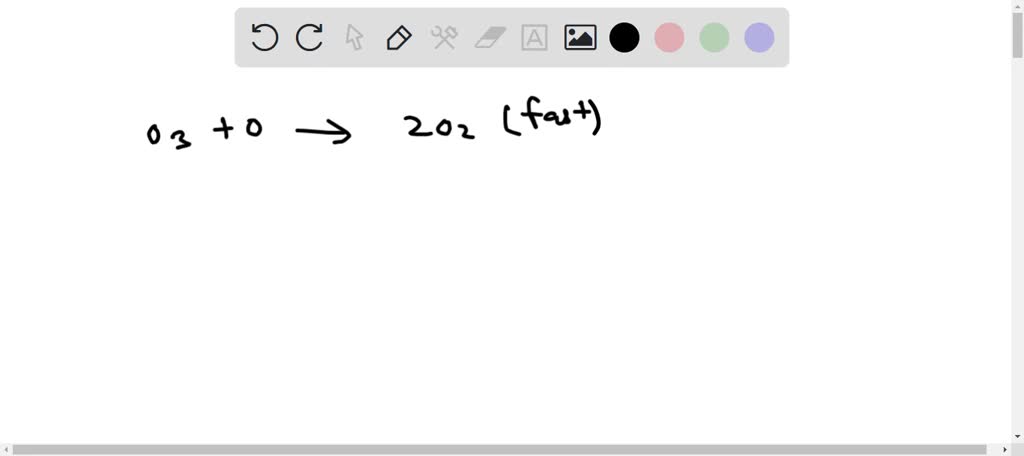
SOLVED: The decomposition of ozone, O3, is believed to occur by the two-step mechanism O3 → O2 + O (slow) O + O3 → 2O2 (fast) 2O3 → 3O2 (net reaction) If

Ozone, O3, Trioxygen, Inorganic Molecule. it is an Allotrope of Oxygen Stock Vector - Illustration of dioxygen, molecule: 197174422
![The ozone, O3, of the stratosphere can be decomposed by reaction with nitrogen oxide (commonly called nitric oxide),NO, from high-flying jet aircraft. The rate expression is rate = k[O3][NO]. Which of the The ozone, O3, of the stratosphere can be decomposed by reaction with nitrogen oxide (commonly called nitric oxide),NO, from high-flying jet aircraft. The rate expression is rate = k[O3][NO]. Which of the](https://homework.study.com/cimages/multimages/16/7133470169922890101680733.png)
The ozone, O3, of the stratosphere can be decomposed by reaction with nitrogen oxide (commonly called nitric oxide),NO, from high-flying jet aircraft. The rate expression is rate = k[O3][NO]. Which of the





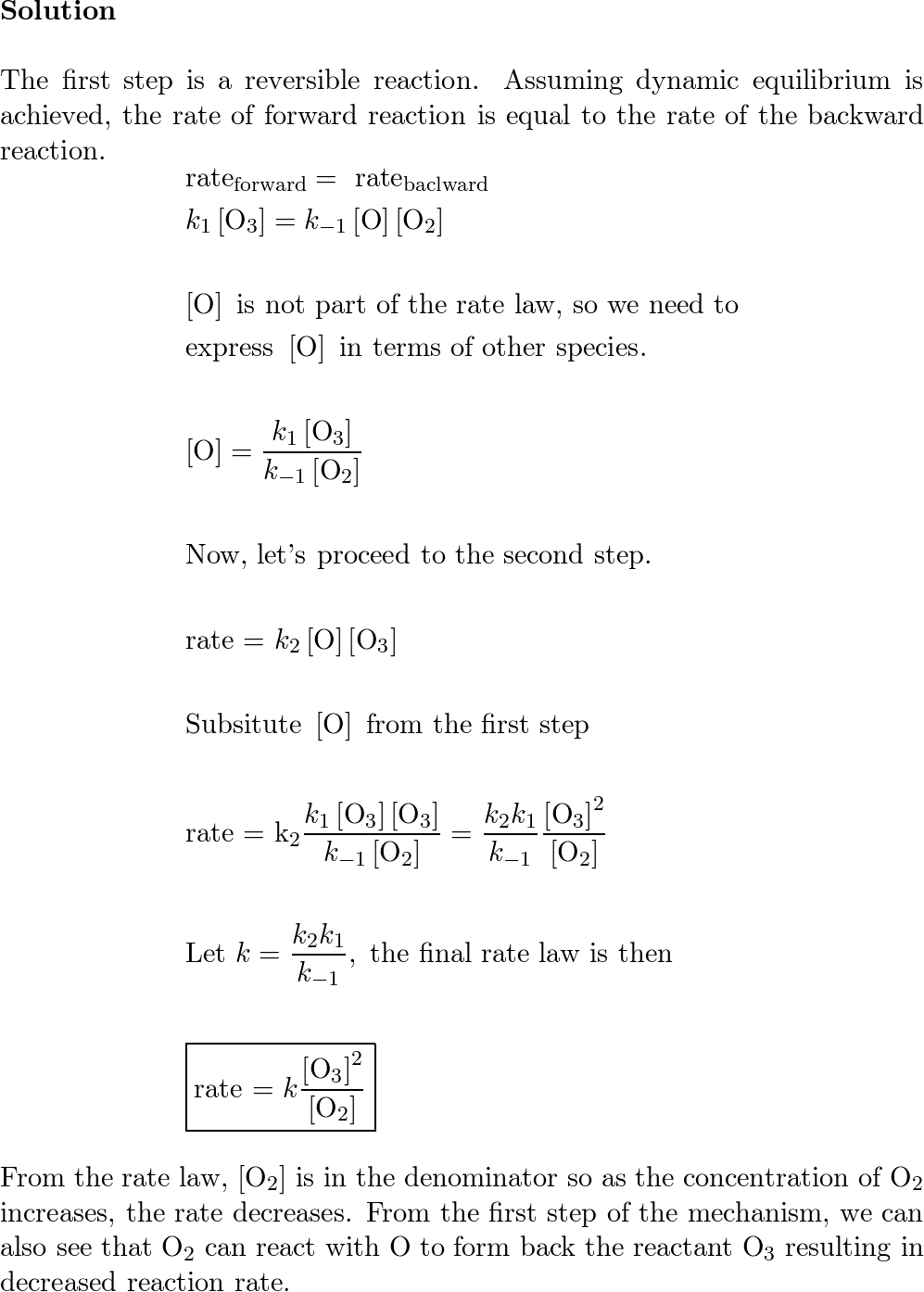
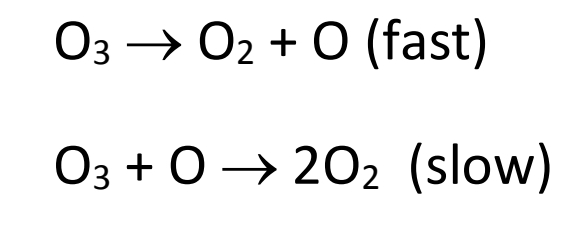
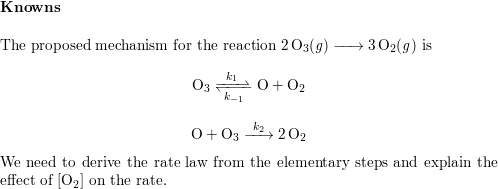


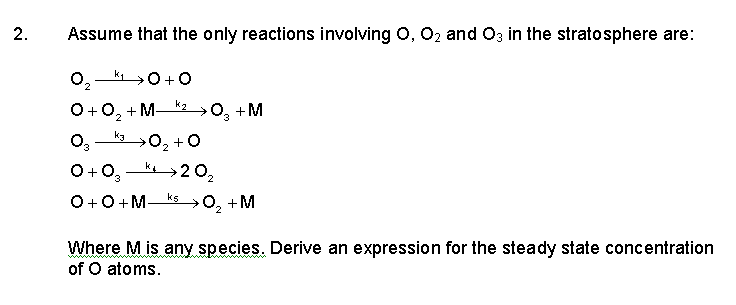
![The [O III]λ5007 EW versus the O3/O2 ratio measured from the VIRUS... | Download Scientific Diagram The [O III]λ5007 EW versus the O3/O2 ratio measured from the VIRUS... | Download Scientific Diagram](https://www.researchgate.net/publication/351448886/figure/fig3/AS:1021754944270337@1620616770835/The-O-IIIl5007-EW-versus-the-O3-O2-ratio-measured-from-the-VIRUS-spectra-for-our.png)


