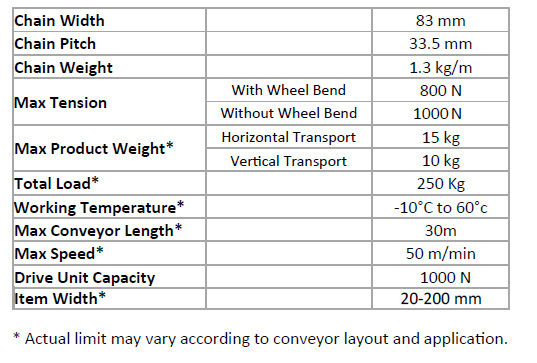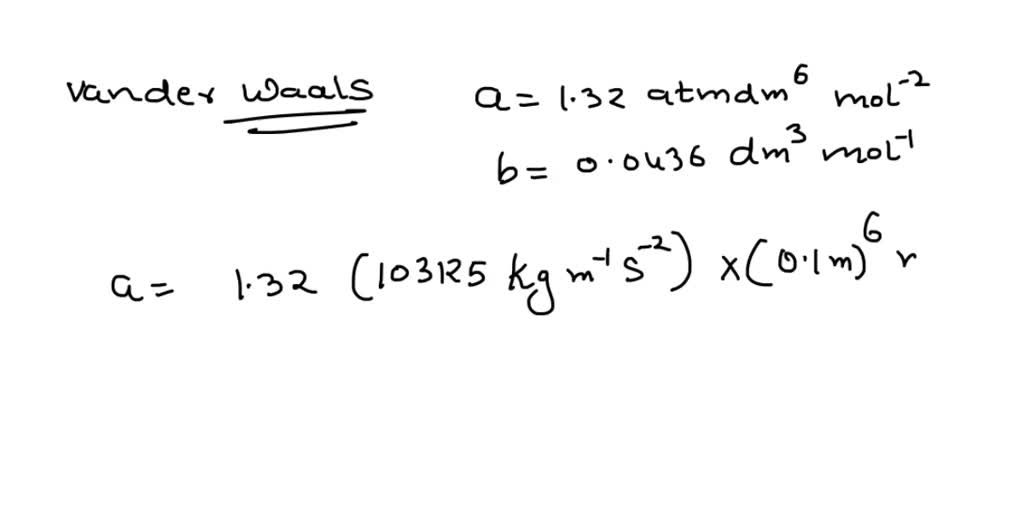
SOLVED: One mole of a van der Waals gas at 300 K expands isothermally and reversibly from 20 dm3 to 60 dm3 (a = 0.556 m6 Pa mol-2 ; b = 0.064

Payload is defined as the difference between the mass of displaced air and the mass of the balloon. Calculate the payload, when a balloon of radius 10 m of mass 100 kg
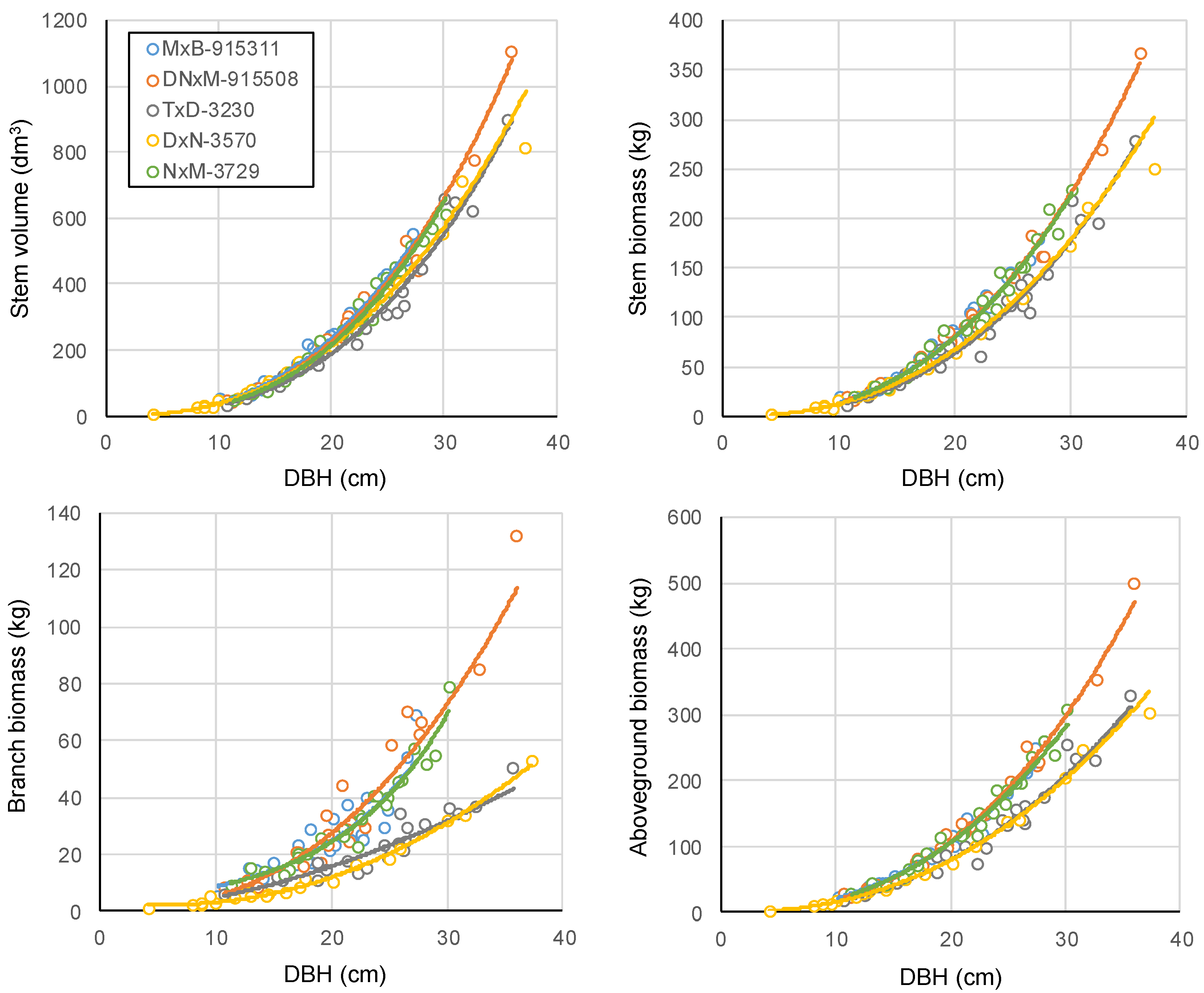
Forests | Free Full-Text | Biomass and Volume Yield in Mature Hybrid Poplar Plantations on Temperate Abandoned Farmland

SOLVED: In a 0.300 mol/kg aqueous solution of sucrose, the C12H22O11 molarity is 0.200 mol/dm3 at 20°C and 1 atm. The density of water is 0.998 g/cm3 at 20°C and 1 atm. (

SOLVED: To what temperature can a 20 dm3 oxygen pressure flask containing 1.6 kg of oxygen be heated if the highest possible pressure is 152 bar? The van der Waals constants for

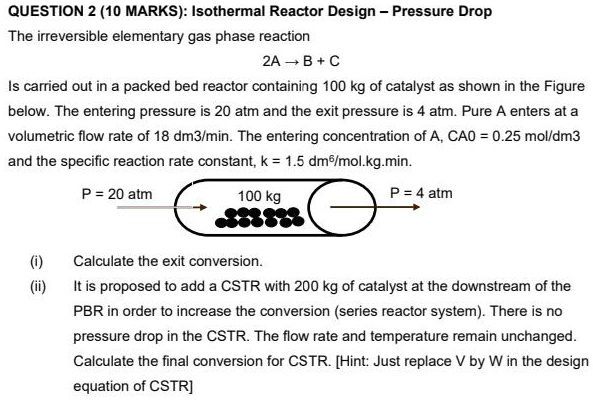
SOLVED: P11-4A The elementary irreversible gas-phase reaction A>B+C is carried out adiabatically in a PFR packed with a catalyst. Pure A enters the reactor at a volumetric flow rate of 20 dm3/s,

Mettler Toledo Analytical Weight Sets: ASTM Class 1:Balances and Scales:Calibration | Fisher Scientific

1959-sss/DM3 elicits dose-dependent antitumor activity. Tumor growth... | Download Scientific Diagram